Author: Sarah Daniel

Globally, the absolute number of people falling ill with TB decreased in 2024 for the first time since 2020, following 3 consecutive years of increases (2021–2023) due to COVID related disruptions to TB diagnosis and treatment. The total of 10.7 million was a small (1%) reduction from 10.8 million in 2023, but still above the level of 2020 (10.3 million).[12] There was a larger (1.7%) global decline in the TB incidence rate between 2023 and 2024; at 131 per 100 000 population in 2024, this was back to the level of 2020. The net reduction from 2015 to 2024 was 12%, far from the End TB Strategy milestone of a 50% reduction by 2025 and the target of an 80% reduction by 2030.[12] Globally, the number of deaths caused by TB also fell in 2024. The total of 1.23 million was a 3% reduction compared with 1.27 million in 2023. The net reduction from 2015 to 2024 is more impressive, at 29%, but still far from the End TB Strategy milestone of a 75% reduction by 2025 and the target of a 90% reduction by 2030[12].
Despite being preventable and curable, tuberculosis remains one of the world’s deadliest infectious diseases. A key reason is not just gaps in treatment, but gaps in data. Modern Tuberculosis surveillance; defined as the continuous, systematic collection, analysis and use of individual case and laboratory data, enables faster case finding, more effective treatment monitoring, and better resource allocation. The World Health Organisation’s 2024 surveillance guidance emphasises unified, digital, case-based systems across the entire care pathway [1][2]. Countries using such systems can link lab networks, digital registries and community screening data to detect outbreaks and drug-resistant cases in near real-time. For example, Nigeria’s use of an Early Warning Outbreak Recognition System (EWORS) to map TB case clusters led to higher case detection yields in “hotspot” areas and more efficient screening[3]. India’s integrated digital platform (“Ni-kshay”) ties together case notification, treatment outcomes, adherence and patient support, accelerating response efforts[4]. South Africa has even launched a near-real-time public TB dashboard to inform policy and track progress towards testing targets[5]. While these advances are significant, challenges remain including incomplete data, interoperability gaps, and concerns around patientprivacy, requiring sustained investment, workforce training, and strong data-governance frameworks. Nonetheless, when well-implemented, robust surveillance measurably improves TB outcomes (e.g. driving up treatment success rates to ~88% globally[7]) and brings us closer to End-TB goals.

What is TB Surveillance? Definition and Components
Tuberculosis surveillance is the continuous, systematic collection, analysis and reporting of data related to TB infection and disease in the population[2]. Core components include case notification (individual records of diagnosed TB cases, drug-susceptibility results, HIV status, etc.), treatment registries (to track outcomes), laboratory networks (culture and drug-resistance testing data), and mortality data. In practice, surveillance draws on multiple data sources including facility case registers, laboratory databases (e.g. GeneXpert logs), contact-investigation records, and community screening activities which feed into a national system.
The WHO provides standardized definitions and forms to ensure consistency across countries. Historically, WHO’s guidance on TB recording and reporting (first issued in the mid-1990s and updated in 2006 and 2013[2]) set the stage for the Global TB Report and annual country data collection.
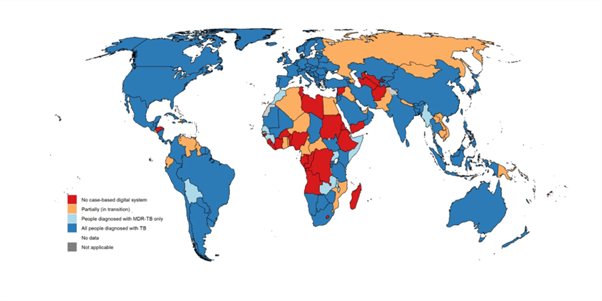
Surveillance Improves Case Detection
Quality surveillance drives case finding. When routine data identify areas or groups with low detection, programs can target them for active case finding. For example, spatial analysis of surveillance data can reveal “hotspots” of transmission. In one Nigerian project, TB notification data were fed into an Early Warning Outbreak Recognition System (EWORS) to map case clusters at the ward level. Subsequent targeted screening in EWORS-identified hotspots yielded significantly more cases than screening in non-hotspot areas[3]. In 14 high-burden Nigerian states, the number of presumptive cases needed to detect one TB case fell (from about 14 per 10,000 to 10 per 10,000) in hotspots[3]. In other words, data-driven targeting increased the yield and precision of active case finding by over 33%. Similarly, retrospective analyses in low-burden settings (e.g. Japan) have shown that routine notification data, if monitored continuously, can flag unusual case surges or outbreaks for investigation[9].
Digital, case-based surveillance systems enhance case detection by enabling real-time monitoring. Automated alerts can be built into electronic registers to signal suspected outbreaks or missed notifications. GIS mapping tools overlay TB case data on maps, helping locate underserved communities. Decision-support dashboards allow programs to see where presumptive TB rates are high or where testing coverage is low. For example, WHO and partners have developed DHIS2-based TB data packages that include standard dashboards of core indicators (notifications, testing rates, outcomes) by subnational area[2]. Countries adopting these digital tools can drill down on case detection gaps rapidly. In short, surveillance data illuminate who is being missed and where, enabling strategic screening efforts.
Treatment Monitoring and Outcomes
Surveillance systems also track patients through treatment, which is crucial for monitoring outcomes and preventing relapse or loss to follow-up. Case-based systems ensure that every diagnosed patient is followed until treatment completion (or dropout). Electronic registers can automatically flag late or missing outcomes for follow-up by health workers. This continuous monitoring helps programs maintain high treatment success rates. Indeed, WHO reports that global treatment success for drug-susceptible TB has steadily improved – from 81% in 2016 to 88% in 2023[7] – reflecting better treatment surveillance and adherence support (among other factors). Without robust surveillance, outcome data would be delayed or aggregate, masking suboptimal performance. For drug-resistant TB (DR-TB), surveillance data on treatment enrolment and outcomes are especially critical. Currently only about 43% of estimated MDR/RR-TB cases enter appropriate treatment[10]. Closing that gap requires surveillance-linked interventions: ensuring sputum-positive cases receive rapid drug-susceptibility testing, expanding second-line drug supplies, and strengthening adherence monitoring (e.g. via digital adherence tools). New WHO guidance emphasizes integrating laboratory and clinical data so that all MDR-TB diagnosed can be promptly started on therapy[10].
Drug Resistance Management
Data and surveillance are vital to confronting drug-resistant TB. Laboratory networks must link to surveillance systems so that resistance patterns are reported in real time. Genomic surveillance is increasingly playing a role. Whole-genome sequencing (WGS) of Mycobacterium tuberculosis isolates (once confined to research labs) is now being integrated into public health. The COVID-19 pandemic accelerated genomic capacity in many countries[7]. This “hard-won” investment (next-generation sequencers, bioinformatics training) offers a transformative opportunity: genomic data can rapidly identify resistance-conferring mutations and map transmission chains[7]. For example, detailed strain typing can confirm if MDR-TB cases in a community represent treatment failure or a new outbreak. Leading experts call for global investment in TB genomics to unlock these benefits[7]. In practice, linkage of lab and surveillance data (e.g. via unique patient IDs) ensures that every confirmed DR-TB case feeds into national registers and treatment programs. Strong surveillance also informs resource needs: data on resistance trends guide procurement of newer drugs (bedaquiline, linezolid) and diagnostics (next-gen Xpert). In summary, comprehensive TB surveillance; from phenotypic DST results to genomic data, underpins effective DR-TB control.
Outbreak and Hotspot Detection
Surveillance data serve as an early warning for emerging TB clusters. Public health teams can review trends in notifications (by location, age or setting) to spot unusual spikes. In practice, automated cluster detection algorithms or simple monthly reviews can alert officials to potential outbreaks in congregate settings (e.g. prisons, workplaces) or communities. The Nigeria example above illustrates this at scale: a system designed for outbreak recognition was repurposed to detect TB hotspots for targeted screening[3]. In low-incidence settings, even a handful of cases in a short period may signal transmission. For instance, in Sapporo, Japan, retrospective analysis of ward-level TB rates identified six possible outbreaks over 17 years, two of which were confirmed by local investigators[9]. Similar methods are used in the US, where the CDC’s TB genotyping and cluster reports can trigger local investigations. In essence, robust surveillance (especially when case data include geo-locations) allows programs to detect and respond to TB clusters before they grow.
Resource Allocation and Planning
Data-driven surveillance informs how programs allocate resources. By identifying high-burden or under-served areas, TB programs can prioritize where to deploy mobile X-ray units, GeneXpert machines, or community health workers. WHO urges countries to use surveillance data for strategic planning and priority setting[11]. A recent WHO policy brief notes that “data-driven, people-centered planning” is essential for maximizing impact under tight budgets[11]. For example, if surveillance shows a district with persistently low case detection but high estimated incidence, funds can be directed toward intensified case finding there. Conversely, areas with stable low incidence might shift focus toward prevention (TB infection screening, preventive therapy). In pandemic response and beyond, countries have seen the value of dashboards: South Africa’s new near-real-time TB dashboard (launched 2025) will publicly display testing and case data by province/district[6]. Policymakers and health managers can use this to allocate staff or funding dynamically, track performance towards goals (e.g. five million tests), and improve accountability.
Digital Case-Based Surveillance Systems
Moving from paper registries to unified digital systems is a cornerstone of modern TB surveillance. Digital, case-based systems offer numerous advantages: automated data quality checks, timely access to individual-level data, and flexible analysis by patient age, sex and location[2]. The WHO Global TB Programme and partners have supported DHIS2-based packages for TB data. Since 2019, WHO published a digital data package for aggregate TB reporting (for countries not yet ready for case-based) and, by late 2020, a unified case-based DHIS2 package covering both drug-susceptible and drug-resistant TB[2]. These packages include machine-readable configurations, core indicators and standard dashboards (notifications, testing rates, outcomes) for immediate use[2]. As of 2021, 130 countries had implemented a national digital, case-based TB surveillance system covering all diagnosed patients[2], while others were transitioning from mixed paper-digital systems[2]. For example, India’s Ni-kshay system is a comprehensive digital TB information platform. Ni-kshay integrates case notification from both public and private providers, treatment registries, laboratory results, and patient support (including cash transfers and adherence tools) into one system[4]. This real-time information flow has “accelerated the TB response in India” by enabling follow-up actions (e.g. tracing missing patients, delivering incentives) without delay[4]. Digital systems also facilitate interoperability: by adopting WHO data standards, new tools (mobile apps, lab networks) can link into the same national database.
Laboratory Networks and Connectivity
A robust TB surveillance system relies on strong laboratory networks. All diagnostic labs (microscopy, GeneXpert, culture) should report results into the national database. Remote connectivity solutions (e.g. e-Labs, GeneXpert connectivity software) are increasingly used so that test results flow electronically from peripheral sites to central systems. The WHO’s Supranational Reference Laboratory Network (SRLN) provides quality assurance and links specialised labs globally. Networked labs ensure that if a patient is diagnosed at one site, their data (including drug susceptibility) appear in the surveillance registry, enabling treatment teams to act. Moreover, connectivity allows programs to monitor equipment utilization and supply chains (e.g. cartridge stockouts) as part of surveillance. In practice, countries expanding Xpert access often deploy digital tools simultaneously, so that each new machine reports data automatically into the TB database. These data help monitor if lab expansion is translating into increased TB detection and drug-resistance case finding.
Genomic Surveillance
Genomic tools are an emerging layer of TB surveillance. Whole-genome sequencing (WGS) of M. tuberculosis isolates can identify transmission links and predict drug-resistance mutations. The COVID-19 pandemic left many public health labs in low- and middle-income countries with new sequencing capacity and expertise[7]. TB programs can leverage this “hard-won capacity” to integrate WGS into routine surveillance[7]. For example, sequencing can rapidly distinguish re-infection from treatment failure or confirm a suspected MDR-TB cluster. In Latin America, genotyping has already revealed distinct regional strain lineages, guiding targeted interventions. Looking ahead, combining genomic data with machine learning and clinical records holds promise: algorithms may one day predict drug resistance or transmission hotspots before phenotypic tests complete[7]. However, implementing TB genomics at scale requires careful planning (costs, bioinformatics, data storage) and should complement, not replace, traditional surveillance.
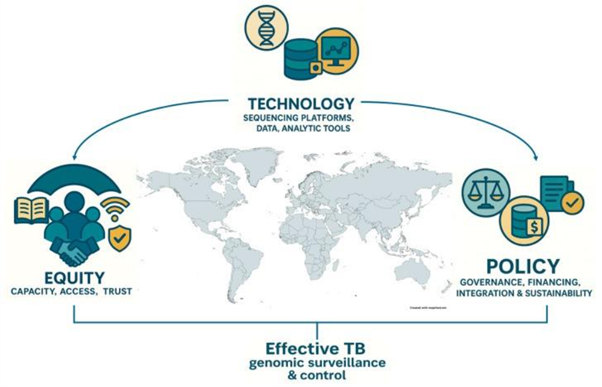
Contact Tracing and Community Data
Contact investigation is both a form of surveillance and an intervention. Recording contact data (who was exposed to a TB case, their test results, and treatments) should feed into the surveillance system. Digital contact-management tools can prompt health workers to screen and follow up all household contacts, ensuring these cases are captured in the data. Community-based active case finding (in prisons, mines, slums) also generates surveillance data: mobile screening vans and NGOs often record suspected TB cases in field registers. Increasingly, these data are digitized (via mobile apps like CommCare) and synced with national systems. For example, Nigeria’s EWORS project pulled “real-time data from a CommCare-based mobile app” on routine TB cases in facilities, using it daily to predict community hotspots[3]. Such community-level data can fill gaps where health facility data are incomplete, improving the overall surveillance picture.
Implementation Challenges and Solutions
Infrastructure and Connectivity: Many high-burden countries still struggle with unreliable electricity and internet in remote clinics. Digital surveillance requires computers or tablets at peripheral sites and regular data uploads. Investments in basic infrastructure (solar power, mobile networks) and offline-capable tools can mitigate this. For example, DHIS2 apps allow data entry offline and syncing when connectivity resumes.
Data Quality: Incomplete or inaccurate data undermine surveillance. Common issues include duplicate records, missing entries, and misclassification. Digital systems help by enforcing data validation rules (e.g. requiring mandatory fields). Regular data audits and feedback loops (data review meetings) are necessary. WHO’s 2024 surveillance guidance includes checklists and benchmarks for data quality, as well as advice on linking records (e.g. to find unreported cases [1].
Workforce and Training: Implementing new systems demands trained staff. Data clerks, lab technicians and epidemiologists need instruction on using electronic registers and interpreting dashboards. WHO and partners offer training modules (e.g. the “TB digital” exercise books accompanying the DHIS2 packages[2]). Continual training (including digital literacy for front-line health workers) is essential.
Funding and Sustainability: Sustained financing is needed to maintain and upgrade surveillance platforms. Partnerships (Global Fund, bilateral donors) can support initial roll-outs, but countries should plan for domestic funding or incorporate TB surveillance into broader health budgets. The Global Fund’s digital health strategies have backed DHIS2 adoption in many TB programs. Continual advocacy for the importance of surveillance is vital to secure this investment.
Interoperability: Many countries have multiple health information systems (for HIV, malaria, general health). TB data should ideally link with these (and with civil registration/vital statistics) to capture TB deaths and co-infections. Adoption of standard data formats (WHO metadata standards) and unique identifiers for patients can facilitate linkage. The development of software-agnostic “digital accelerators” and use of open-source platforms (DHIS2, CommCare, OpenMRS) aim to ease integration[2].
Measurable Impact and Examples
- Case Finding Efficiency: As noted, Nigeria’s EWORS approach increased screening efficiency: the number needed to screen per case fell from 14 to 10 per 10,000 in hotspots[3]. This means fewer resources are used per diagnosed case.
- Treatment Outcomes: Robust monitoring has coincided with improved treatment success. WHO reported a rise in global success rates from 81% (2016) to 88% (2023)[6], suggesting better program management and patient support (to which surveillance contributes).
- Drug-Resistance Control: In countries scaling up genotypic testing and digital recording, the share of TB patients tested for rifampicin resistance has grown. For instance, the Global TB Report notes that gaps between diagnosed MDR-TB cases and enrolled treatment have narrowed year by year[10]. While still only ~43% of MDR/RR-TB cases receive treatment[10], surveillance data now allow national programs to pinpoint where diagnostic and treatment access must improve.
- Transparency and Accountability: Public dashboards (as in South Africa[5]) allow civil society and media to hold programs to account. When data are transparent, funds and efforts align better with epidemiological need. Early experience suggests that communities engaged with data (e.g. civil-society groups following district notifications) contribute to stronger case-finding campaigns.
Conclusion
Effective TB control depends on strong surveillance. By transforming raw data into actionable intelligence (from detecting undiagnosed cases to guiding treatment of drug-resistant TB) surveillance systems materially boost program performance. The shift to digital, case-based platforms (with linked lab, genomic and community data) has been accelerated by WHO guidance and global partnerships[2][1]. High-burden countries are already seeing benefits: higher case finding yields, more timely outbreak response, and improved treatment metrics[3][6]. Moving forward, investments in IT infrastructure, workforce capacity, and data governance will be needed to sustain and expand these gains. With continued innovation and ethical data use, surveillance will remain a cornerstone for reaching End TB targets.
References:
- WHO. WHO releases new guidance on Tuberculosis surveillance. News release, 29 May 2024. (Defines TB surveillance, emphasizes digital case-based systems)[1]
- WHO. Global TB Report 2021: Featured Topic – Case-based TB surveillance. (Overview of TB surveillance evolution, benefits of digital systems)[2].
- Ogbudebe et al. Identifying Hot Spots of Tuberculosis in Nigeria Using an Early Warning Outbreak Recognition System. JMIR Public Health Surveill. 2023;9:e40311. (Case study: EWORS mapping improved TB case detection yield in Nigeria[3]).
- Rao et al. Lessons and updates from India’s National TB Elimination Program: fast-tracking progress toward ending TB. Int J Infect Dis. 2025. (India’s Ni-kshay digital system and innovations for TB response[4].
- South African Government, Health Minister’s Office. Minister Aaron Motsoaledi launches interactive public-facing TB Dashboard, Media Advisory, 24 Oct 2025. (SA launches near-real-time TB data dashboard to support decision-making[5]).
- Mboowa G. Reimagining TB Control in the Era of Genomics. Pathogens 2025;14(10):975. (Calls for integrating whole-genome sequencing into TB surveillance; COVID-19 has built genomic capacity[7]).
- “Ethical Data Governance in Global Pathogen Surveillance”. Prism – Sustainability Journal, 2021. (Discussion of privacy and governance challenges in digital surveillance; notes risk of intrusive data use[8]).
- Can a TB outbreak be detected by reviewing the surveillance data? – PMC[9]
- 2.4 Drug-resistant TB treatment[10]
- Priority setting in tuberculosis programme planning: policy brief [11]
- Global Tuberculosis Report. World Health Organization (WHO). 2025[12]
